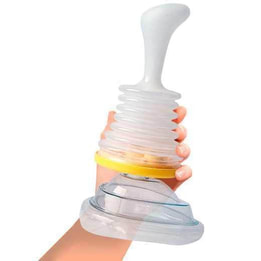
Certification and Research
In Australia, LifeVac has been listed in the ARTG Number 285082
In the USA, LifeVac is registered by the Federal Drug Administration (FDA)
In Canada, LifeVac is licenced as a medical device by Health Canada
In the United Kingdom, LifeVac is registered in by the Medicines and Healthcare Products Regulatory Agency (MHRA) and carries a CE conformity certificate.
Dunne CL, Viguers K, Osman S, Queiroga AC, Szpilman D, Peden AE. A 2-year prospective evaluation of airway clearance devices in foreign body airway obstructions. Resusc Plus. 2023 Nov 8;16:100496. doi: 10.1016/j.resplu.2023.100496. PMID: 38026136; PMCID: PMC10658362.
A study of the LifeVac data concludes that:
1. The data presented here strongly supports its (LifeVac's) utilization in choking emergencies, particularly among at-risk groups like children under the age of five. Incorporating LifeVac as an additional measure can significantly contribute to preventing devastating outcomes in such situations.
2. We urge the resuscitation community to proactively evaluate airway clearance devices to ensure the public remains updated in best practices”
A study of the LifeVac data concludes that:
1. The data presented here strongly supports its (LifeVac's) utilization in choking emergencies, particularly among at-risk groups like children under the age of five. Incorporating LifeVac as an additional measure can significantly contribute to preventing devastating outcomes in such situations.
2. We urge the resuscitation community to proactively evaluate airway clearance devices to ensure the public remains updated in best practices”
Costable, Nicholas J.; Costable, John M.; Rabin, Glenn. The use of LifeVac, a novel airway clearance device, in the assistance of choking victims aged five and under: Results of a retrospective 10-year observational study. Journal of Pediatric Critical Care 11(3):p 93-98, May–Jun 2024. | DOI: 10.4103/jpcc.jpcc_3_24
A analysis of 10-years of usage data of the LifeVac (a larger sample than any research on first-aid measure) concluded that:
1. “LifeVac should be considered a valuable complement to standard life support techniques in choking emergencies, particularly for at-risk groups such as children under the age of 5.”
2. However, when a choking incident does occur, even when the standard protocol is followed (i.e., Heimlich manoeuvre), this fails around 26% of the time in the general population.[11] In addition, it takes several minutes to get first responders on the scene. Brain death can occur within a matter of minutes if the airway is completely obstructed, causing a lack of oxygen to the brain. In general, permanent brain damage can occur within 4–6 min of cardiac arrest caused by choking.[12] This highlights the importance of quickly and effectively addressing a choking emergency to restore airflow and prevent permanent brain damage or death.
3. Although LifeVac is not intended to replace standard choking protocols (Heimlich manoeuvre, back blows, chest thrusts), the data presented here strongly support its utilization in choking emergencies, particularly among at-risk groups like children under the age of five. Incorporating LifeVac as an additional measure can significantly contribute to preventing devastating outcomes in such situations.
A analysis of 10-years of usage data of the LifeVac (a larger sample than any research on first-aid measure) concluded that:
1. “LifeVac should be considered a valuable complement to standard life support techniques in choking emergencies, particularly for at-risk groups such as children under the age of 5.”
2. However, when a choking incident does occur, even when the standard protocol is followed (i.e., Heimlich manoeuvre), this fails around 26% of the time in the general population.[11] In addition, it takes several minutes to get first responders on the scene. Brain death can occur within a matter of minutes if the airway is completely obstructed, causing a lack of oxygen to the brain. In general, permanent brain damage can occur within 4–6 min of cardiac arrest caused by choking.[12] This highlights the importance of quickly and effectively addressing a choking emergency to restore airflow and prevent permanent brain damage or death.
3. Although LifeVac is not intended to replace standard choking protocols (Heimlich manoeuvre, back blows, chest thrusts), the data presented here strongly support its utilization in choking emergencies, particularly among at-risk groups like children under the age of five. Incorporating LifeVac as an additional measure can significantly contribute to preventing devastating outcomes in such situations.
International Study verifies our claims that LifeVac works as designed.
Pre-hospital removal improves neurological outcomes in elderly patient with foreign body airway obstruction
YutakaIgarashiMD, PhD, ShojiYokoboriMD, PhD, YudaiYoshinoMD, TomohikoMasunoMD, PhD, MasatoMiyauchi MD, PhD, HiroyukiYokotaMD, PhD
Abstract
Objective
In Japan, the number of patients with foreign body airway obstruction by food is rapidly increasing with the increase in the population of the elderly and a leading cause of unexpected death. This study aimed to determine the factors that influence prognosis of these patients.
Methods
This is a retrospective single institutional study. A total of 155 patients were included. We collected the variables from the medical records and analysed them to determine the factors associated with patient outcome. Patient outcomes were evaluated using cerebral performance categories (CPCs) when patients were discharged or transferred to other hospitals. A favourable outcome was defined as CPC 1 or 2, and an unfavourable outcome was defined as CPC 3, 4, or 5.
Results
A higher proportion of patients with favourable outcomes than unfavourable outcomes had a witness present at the accident scene (68.8% vs. 44.7%, P = 0.0154). Patients whose foreign body were removed by a bystander at the accident scene had a significantly high rate of favourable outcome than those whose foreign body were removed by emergency medical technicians or emergency physician at the scene (73.7% vs. 31.8%, P < 0.0075) and at the hospital after transfer (73.7% vs. 9.6%, P < 0.0001).
Conclusions
The presence of a witness to the aspiration and removal of the airway obstruction of patients by bystanders at the accident scene improves outcomes in patients with foreign body airway obstruction. When airway obstruction occurs, bystanders should remove foreign bodies immediately.
Objective
In Japan, the number of patients with foreign body airway obstruction by food is rapidly increasing with the increase in the population of the elderly and a leading cause of unexpected death. This study aimed to determine the factors that influence prognosis of these patients.
Methods
This is a retrospective single institutional study. A total of 155 patients were included. We collected the variables from the medical records and analysed them to determine the factors associated with patient outcome. Patient outcomes were evaluated using cerebral performance categories (CPCs) when patients were discharged or transferred to other hospitals. A favourable outcome was defined as CPC 1 or 2, and an unfavourable outcome was defined as CPC 3, 4, or 5.
Results
A higher proportion of patients with favourable outcomes than unfavourable outcomes had a witness present at the accident scene (68.8% vs. 44.7%, P = 0.0154). Patients whose foreign body were removed by a bystander at the accident scene had a significantly high rate of favourable outcome than those whose foreign body were removed by emergency medical technicians or emergency physician at the scene (73.7% vs. 31.8%, P < 0.0075) and at the hospital after transfer (73.7% vs. 9.6%, P < 0.0001).
Conclusions
The presence of a witness to the aspiration and removal of the airway obstruction of patients by bystanders at the accident scene improves outcomes in patients with foreign body airway obstruction. When airway obstruction occurs, bystanders should remove foreign bodies immediately.
The International Liaison Committee on Resuscitation (ILCOR) review the evidence around the relief of upper-airway obstruction, including all methods and devices.
The peak body in the world for resuscitation science has just release a report showing the review of the evidence available for methods and devices used to relieve upper airway obstructions. A summary of the outcomes and recommendations is below. The full text is available on the ILCOR website.
Summary of ILCOR Systematic Review: Removal of Foreign Body Airway Obstruction
The International Liaison Committee on Resuscitation (ILCOR) conducted a systematic review of the evidence surrounding various techniques used for the removal of upper airway obstruction. The techniques included both first aid measures and equipment, including Magill forceps and suction-based airway devices (such as the LifeVac). https://costr.ilcor.org/document/removal-of-foreign-body-airway-obstruction-tfsr-costr
One must remember that whilst the ILCOR is a research body that undertakes a constant review of its treatment recommendations (but no surveillance or monitoring of efficacy, complications, or harm i.e. GRADE review without an evaluation of the desirable and undesirable consequences nor a post-implementation review); the ARC in Australia/New Zealand is a non-government, voluntary, member-representative organisation that constructs local opinion-based guidelines i.e. Good Practice Statements*.
*Definition: Good practice statements. Guideline panels often consider it necessary to issue guidance on specific topics that do not lend themselves to a formal review of research evidence. The reason might be that research into the topic is unlikely to be located or would be considered unethical or infeasible. Criteria for issuing a nongraded good practice statement include the following: There is overwhelming certainty that the benefits of the recommended guidance will outweigh the harms, and a specific rationale is provided; the statements should be clear and actionable to a specific target population; the guidance is deemed necessary and might be overlooked by some providers if not specifically communicated; and the recommendations should be readily implementable by the specific target audience to which the guidance is directed.
It should be noted that the primary criteria for the use of these non-graded practice statements is that ‘there is overwhelming certainty that the benefits of the recommended guidance will outweigh the harms’. This bar has certainly not been reached in the ANZCOR choking recommendations/ guidelines and it is clear that, unlike device regulators, resuscitation bodies do not operate under a risk management methodology that looks at harm.
“Good practice statements are not recommendations but represent expert opinion in light of very limited data.” These can be divergent from the ILCOR treatment recommendations and the evidence if there are local factors or an audience that requires differing evidence-based advice e.g. venomous bites in Australia. This should not result in divergent recommendations that lack sufficient evidence. While the ANZCOR guidelines claim to be “based on the scientific evidence, clinical experience, community values, and good sense in applying the guideline.” The resultant guidelines “may be made independent of the levels of evidence.”, “may not be as effective as other techniques” and “may not be appropriate in every circumstance”. While these are acknowledged by the ARC, the poor processes, lack of peer review, and lack of good sense are not a rationale for having divergent and unevidenced guidelines. This was never the intent of the ILCOR.
There has been much more usage (over 1900 saves), research, and case series data concerning the LifeVac™ since this review by the ILCOR. Usage reports to regulators worldwide have shown a high level of efficacy and safety, with no reports of failure, harm, or complications from its use at any age. Further studies have shown superior efficacy to existing measures (including abdominal thrusts) and ease of use rated as higher than first-aid measures. So far none of these non-regulatory resuscitation organisations, despite claims to be ‘waiting for more evidence’, have not been able to describe any such study that would reach their threshold of ‘evidence’ for the LifeVac™. As there is no pilot or evaluation process for devices (or first aid measures), this will result in an indefinite freeze on life-saving innovation.
The International Liaison Committee on Resuscitation (ILCOR) conducted a systematic review of the evidence surrounding various techniques used for the removal of upper airway obstruction. The techniques included both first aid measures and equipment, including Magill forceps and suction-based airway devices (such as the LifeVac). https://costr.ilcor.org/document/removal-of-foreign-body-airway-obstruction-tfsr-costr
- It is the opinion of ILCOR that foreign body airway obstruction (FBAO) is a common problem and many cases are likely to be resolved easily, without the need to involve healthcare providers. ILCOR did recognise that FBAO is however an important cause of early mortality that typically affects the young and old, or individuals with impaired neurological function/swallowing. Further current strategies to remove FBAO are well known to many people, but all interventions can cause harm that may lead to death, as well as delays in treatment (of any kind). Therefore, there is a need to carefully balance the risks and benefits of strategies for removing foreign body airway obstructions (by any means).
- Overall, the ILCOR found that all the evidence on choking management is rated as very low quality (via the GRADE methodology they use) for all outcomes primarily due to a very serious risk of bias due to confounding data. Because of this and a high degree of heterogeneity, no meta-analyses could be performed and they found all individual studies to be difficult to interpret. This was the case regardless of whether the studies concerned first-aid measures or suction-based airway clearance devices.
- ILCOR found evidence of harm has been reported for strategies of back blows, abdominal thrusts, chest thrusts, and blind finger sweeps but no case reports of harm were identified concerning Magill forceps or suction-based airway clearance devices.
- That in recent years, manual suction devices (airway clearance devices) have been developed but these devices have not previously been considered by ILCOR.
- ILCOR noted a higher level of risk with airway clearance devices that incorporate a plastic tube that is inserted into the mouth (e.g. DeChoker), which could conceivably cause harm in a similar way to a blind finger sweep and that further evidence on safety is required.
- ILCOR found that there are reported cases of benefit for back blows, chest thrusts/ compressions, abdominal thrusts, Magill forceps, finger sweeps, and suction-based airway clearance devices. They also reported cases of harm for all interventions (except suction-based devices) i.e. back blows, chest thrusts, abdominal thrusts, and blind finger sweeps.
- The task force acknowledges that there is some data from a case series demonstrating the efficacy of suction-based airway clearance devices.
- At this time the ILCOR felt that the data were insufficient to support the implementation of a new technology with an associated financial cost. This reflects the primary function of treatment recommendations at ILCOR is to focus on measures of first aid i.e. that can be implemented by anyone without specialised or additional equipment and is therefore outside their scope.
- The ILCOR noted regarding suction-based devices, that the limited number of cases is likely insufficient to provide preliminary data on harm. On this basis, the task force felt that there was insufficient evidence to make a treatment recommendation about these devices. The task force has outlined recommendations for further research about these devices. The format and detail of this data reflect the detail already collected by LifeVac in post-market surveillance. i.e. accurately describe the incidence of FBAO, patient demographics (age, setting, comorbidities, food type, conscious level), full range of interventions delivered, who delivered interventions (health professional/ lay responder), success rates of interventions, harm of interventions, and outcomes. The ILCOR has noted in their report what LifeVac has been asserting for some time i.e. that it is unlikely that such a study can be conducted using only health service data.
- The ILCOR believes that there is a need for further evidence on the benefits and harms of suction-based airway clearance devices and suggested the prospective registration of all device uses and published case series. This has always been the intention of LifeVac. Importantly ILCOR made no comment regarding the role of regulators of medical devices or the databases that they hold of reports from all sources, of use, any failures, complications, or harm. Presumably this is because neither ILCOR nor ANZCOR utilise a risk management framework or understand medical device regulation.
- The treatment recommendations made by the ILCOR based on currently available evidence, re-enforce the guidance provided by LifeVac in its IFU and further discredit the divergent opinions of the Australian Resuscitation Council (ANZCOR). In particular, that ILCOR considered “chest thrusts” a measure for unconscious patients i.e. CPR compressions supine on a firm surface:
- We suggest that back slaps are used initially in patients with a FBAO and an ineffective cough (weak recommendation, very low certainty of evidence).
- We suggest that abdominal thrusts are used in adults and children with a FBAO and an ineffective cough where back slaps are ineffective (weak recommendation, very low certainty of evidence).
- We suggest that rescuers consider the manual extraction of visible items in the mouth (weak recommendation, very low certainty of evidence).
- We suggest against the use of blind finger sweeps in patients with a FBAO (weak recommendation, very low certainty of evidence).
- We suggest that appropriately skilled individuals consider the use of Magill forceps to remove FBAO in OHCA patients with a FBAO (weak recommendation, very low certainty of evidence).
- We suggest that chest thrusts are used in unconscious patients with a FBAO (weak recommendation, very low certainty of evidence).
- We suggest that bystanders undertake interventions to support FBAO removal as soon as possible after recognition (weak recommendation, very low certainty of evidence).
One must remember that whilst the ILCOR is a research body that undertakes a constant review of its treatment recommendations (but no surveillance or monitoring of efficacy, complications, or harm i.e. GRADE review without an evaluation of the desirable and undesirable consequences nor a post-implementation review); the ARC in Australia/New Zealand is a non-government, voluntary, member-representative organisation that constructs local opinion-based guidelines i.e. Good Practice Statements*.
*Definition: Good practice statements. Guideline panels often consider it necessary to issue guidance on specific topics that do not lend themselves to a formal review of research evidence. The reason might be that research into the topic is unlikely to be located or would be considered unethical or infeasible. Criteria for issuing a nongraded good practice statement include the following: There is overwhelming certainty that the benefits of the recommended guidance will outweigh the harms, and a specific rationale is provided; the statements should be clear and actionable to a specific target population; the guidance is deemed necessary and might be overlooked by some providers if not specifically communicated; and the recommendations should be readily implementable by the specific target audience to which the guidance is directed.
It should be noted that the primary criteria for the use of these non-graded practice statements is that ‘there is overwhelming certainty that the benefits of the recommended guidance will outweigh the harms’. This bar has certainly not been reached in the ANZCOR choking recommendations/ guidelines and it is clear that, unlike device regulators, resuscitation bodies do not operate under a risk management methodology that looks at harm.
“Good practice statements are not recommendations but represent expert opinion in light of very limited data.” These can be divergent from the ILCOR treatment recommendations and the evidence if there are local factors or an audience that requires differing evidence-based advice e.g. venomous bites in Australia. This should not result in divergent recommendations that lack sufficient evidence. While the ANZCOR guidelines claim to be “based on the scientific evidence, clinical experience, community values, and good sense in applying the guideline.” The resultant guidelines “may be made independent of the levels of evidence.”, “may not be as effective as other techniques” and “may not be appropriate in every circumstance”. While these are acknowledged by the ARC, the poor processes, lack of peer review, and lack of good sense are not a rationale for having divergent and unevidenced guidelines. This was never the intent of the ILCOR.
There has been much more usage (over 1900 saves), research, and case series data concerning the LifeVac™ since this review by the ILCOR. Usage reports to regulators worldwide have shown a high level of efficacy and safety, with no reports of failure, harm, or complications from its use at any age. Further studies have shown superior efficacy to existing measures (including abdominal thrusts) and ease of use rated as higher than first-aid measures. So far none of these non-regulatory resuscitation organisations, despite claims to be ‘waiting for more evidence’, have not been able to describe any such study that would reach their threshold of ‘evidence’ for the LifeVac™. As there is no pilot or evaluation process for devices (or first aid measures), this will result in an indefinite freeze on life-saving innovation.